  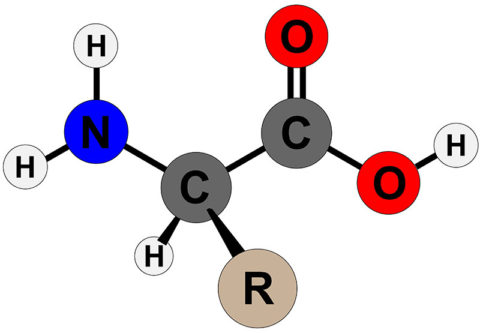
The -NH2 cluster is the stronger base, and then it picks up H+ from the -COOH cluster to get rid of a zwitterion. Amino acids are the known samples of zwitterions. The net charge of the whole molecule is zero. Peptide bond formation: Amino acids will connect with a peptide bond involving their amino and carboxyl groups.Ĭhemical Properties Zwitterionic propertyĪ zwitterion could be a molecule with purposeful groups, of which a minimum of one features a positive and one features a negative electrical charge. 
All amino acids (except glycine) are optically active. On heating to high temperatures, they decompose. R-group of amino acids and pH scale of the solvent plays a vital role in insolubility. Solubility: They’re soluble in water, slightly soluble in alcohol, and dissolve with issues in methyl alcohol, ethanol, and propyl alcohol.Properties of Amino acids Physical PropertiesĪmino acids are colorless, crystalline solid.Īll amino acids have a high M.P(melting point) larger than 200°. Some are residues change when a macromolecule has been synthesized by post-translational modifications others are amino acids present in living organisms however not as constituents of proteins. Alternative amino acids are changed amino acids and are known as non-protein amino acids. 
Over three hundred amino acids are found in nature however solely twenty amino acids are standard and present in protein as a result of they’re coded by genes. Thus, amino acids are the essential unit of proteins. Proteins like amino acids are biosynthesized from twenty amino acids in a very system involving strict genetic management. Amino Acids as ProteinĪmino acids represent a bunch of neutral products clearly distinguished from alternative natural compounds chemically, chiefly attributable to their ampholytic properties, and biochemically, chiefly attributable to their role as protein constituents. over five hundred present amino acids are well-known to represent chemical compound units of peptides, as well as proteins. The element present in each amino acid are carbon (C), H (H), oxygen (O), and gas (N) additionally sulfur (S) is present in the aspect chains of amino acid and essential amino acid, and element (Se) within the less common amino acid selenocysteine. Amino acids are organic compounds that contain amino and carboxyl, functional groups, along with a side chain (R group) specific to every amino acid. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
